CITA ESTE TRABAJO
Sánchez Torrijos Y, Lucena Valera A, Ampuero Herrojo J. Precisión diagnóstica y limitaciones de los test no invasivos en pacientes con esteatosis hepática metabólica. RAPD Online. 2021;44(5):168-172.
Introduction
Metabolic-associated fatty liver disease (MAFLD) is a clinical condition characterized by the deposition of fat in the liver, accompanied by an inflammatory component (non-alcoholic steatohepatitis (NASH)) and, finally, hepatic fibrosis1,2 in the absence of other etiologies. Currently, probably influenced by the current lifestyle, the prevalence of MAFLD is about 25-30% in the general population3,4 and more elevated in patients showing metabolic features, such as obesity and diabetes mellitus. In fact, the European Association for the Study of the Liver5 recommends screening for MAFLD in the presence of insulin resistance or metabolic syndrome. Although a small proportion of patients suffers from an advanced MAFLD, due to its high prevalence, it represents the first cause of liver disease in the Western world6 and the second indication for liver transplantation in the USA7.
The liver biopsy is the gold standard to detect and stage MAFLD. However, given the significant number of at-risk patients and the inherent limitations of the biopsy (invasiveness, poor acceptability, sampling variability, and cost), there is an unmet need to find non-invasive and cost-effective tests for the diagnosis and staging of MAFLD. Non-invasive methods should aim to diagnose patients with MAFLD and, on the other hand, identify those with a worse prognosis and/or at-risk of disease progression. They should also be helpful for the subsequent monitoring and assessment of the response to the treatment. Of note, although the levels of alanine aminotransferase (ALT) and aspartate aminotransferase (AST) may be elevated up to four times in MAFLD patients8, they can be also normal or minimally elevated and do not correlate with the fibrosis stage9. Therefore, their monitoring is not recommended for MAFLD management.
This review aims to revise the different predictive models and non-invasive tests (NITs) used to detect fibrosis in the MAFLD scenario, including their limitations in clinical practice.
The spectrum of MAFLD and the use of non-invasive tests
The diagnosis of MAFLD is based on detecting steatosis defined as fat in ≥5% of hepatocytes by histology10, in the absence of a relevant alcohol intake. There are currently several scores that assess hepatic steatosis based on epidemiological, biochemical, and anthropometric data. For example, fatty liver index (FLI), hepatic steatosis index (HSI), and SteatoTest® have been validated externally in the general population showing an optimal accuracy for detecting steatosis (AUROC>0.80)11,12. The main advantages of these scores are their wide availability, their easy implementation and interpretation, and the low cost. However, its usefulness is limited due to the low prognostic implication.
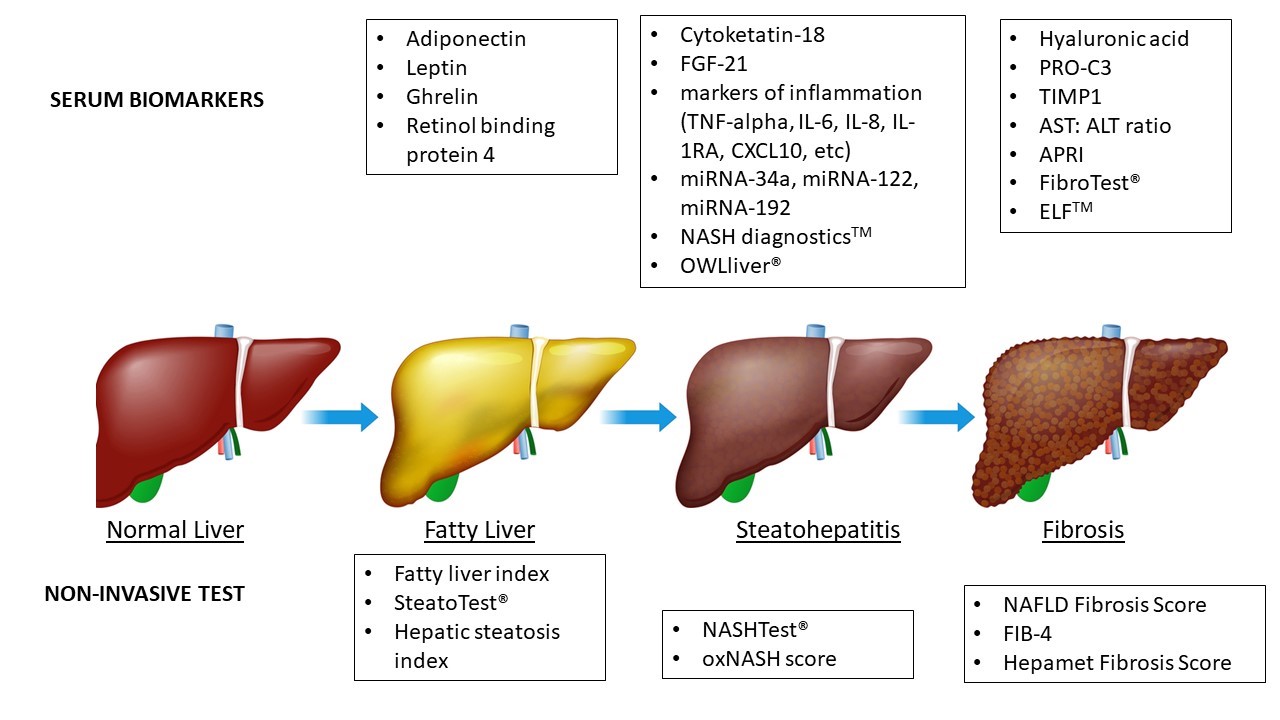
On the other hand, NASH is characterized by the presence of steatosis, hepatocellular damage (e.g., ballooning, apoptosis, necrosis), and lobular inflammatory infiltration13. The diagnosis of NASH provides important prognostic information and indicates an increased risk of progression to fibrosis, cirrhosis, and extrahepatic events13. Non-invasive diagnosis of NASH represents a current challenge, opposite to steatosis and fibrosis, since there is a lack of non-patented NITs showing optimal results. NASHTest® is an algorithm consisting of a combination of age, sex, height, weight, and serum levels of triglycerides, cholesterol, alpha 2 macroglobulin, apolipoprotein AI, haptoglobin, GGT, ALT, AST, and total bilirubin. The AUROC of NASHTest® for the diagnosis of NASH was 0.79 (95%C I 0.69-0.86) for the estimation and 0.79 (95% CI 0.67-0.87) for the validation cohort14. One of the more novel techniques used to identify NASH markers is to detect molecules highly related to NASH using genomic, epigenomic, metabolomic, transcriptomic, or proteomic (-omic approaches)16. OWLiver® is a laboratory test in which different serum lipids and amino acids are measured and combined to define a triglyceride panel that helps differentiate between a normal liver and MAFLD, including distinguishing NASH from a fatty liver. OWLiver® showed an AUROC, sensitivity, and specificity of 0.88, 0.94, and 0.57, respectively, for the discrimination between MAFLD and normal liver, and 0.79, 0.70, and 0.81, respectively, for the discrimination between NASH and simple steatosis15. Figure 1 summarizes the spectrum of NAFLD and the main NITs stage by stage.
Figura 1
Serum biomarkers and non-invasive tests in MAFLD. FGF-21, fibroblast growth factor 21; TIMP1, tissue inhibitor of metalloproteinases 1; CXCL10, chemokine 10; IL-1RA, IL-1 receptor antagonist protein; miRNAs, microRNAs; PRO-C3, a marker of type III collagen formation; APRI, AST: platelet ratio index; ELF, enhanced liver fibrosis.
Finally, fibrosis is the progressive deposition of extracellular matrix in the liver parenchyma that precedes the development of cirrhosis, representing the most important prognostic factor in MAFLD17. Therefore, despite having NITs for the different components of the disease, those detecting fibrosis are more relevant in clinical practice. As mentioned above, although biopsy is the gold standard for diagnosing MAFLD and fibrosis, there is currently a greater acceptance of NITs as an alternative to biopsy, and these are being incorporated into therapeutic and diagnostic algorithms.
The combination of several parameters is considered adequate for the diagnosis of fibrosis in MAFLD. Among non-patented tests, NAFLD Fibrosis Score (NFS), FIB-4, and Hepamet Fibrosis Score (HFS) are the most commonly used19. Both NFS and FIB-4 have been validated externally in MAFLD populations, showing optimal results (AUROC around 0.80)20. For FIB-4, thresholds of 2.67 and 3.25 showed sensitivities of 26.6% and 31.8%, and specificities of 96.5% and 96.0%, respectively. For NFS, a threshold of 1.455 had a sensitivity and a specificity of 72% and 70%, respectively. FS, which includes albumin, platelets, AST, HOMA, age, and sex, has been recently published, showing a significantly higher diagnostic accuracy than NFS and FIB-4 (AUROC 0.85)21. In subsequent studies, HFS has obtained similar AUROCs (0.94 (0.90-0.99, 95% CI) for predicting advanced fibrosis in patients with chronic viral liver disease and MAFLD22. On the other hand, AST:ALT ratio and AST: platelet ratio index (APRI) are simple NITs for detecting advanced fibrosis on MAFLD10, although they were initially derived from chronic hepatitis C cohorts23. Patented or composite biomarkers have demonstrated an improvement in diagnostic precision compared to non-patented scores; however, their application remains limited due to their cost and availability18. For this reason, patented biomarkers are usually reserved for patients with undetermined results for advanced fibrosis after using non-patented NITs16. FibroTest® is a commercially available panel that uses seven parameters, including alpha 2 macroglobulin, haptoglobin, and apolipoprotein‐A1, and showed a high AUROC (0.88) to predict advanced fibrosis in MAFLD24. On the other hand, the enhanced liver fibrosis (ELFTM), which also measures three molecules involved in liver matrix metabolism25, has been used to improve the detection of advanced fibrosis in those with indeterminate fibrosis by other methods26.
Limitations of non-invasive tests in MAFLD
Despite many serum biomarkers and combination panels are published in the literature, there is a lack of independent validation, uncertainty regarding optimum diagnostic cut-off, and a lack of knowledge regarding the influence of other factors that might affect the test interpretation. For instance, although FIB-4, HFS, and NFS have shown promising results, they have some limitations that must be considered (Table 1). In addition, sometimes having low-prevalence populations precludes calculating some operating features of the NITs for advanced fibrosis since, in most studies, the patients who classify initially as low- or high-risk are not biopsied27.
Influence of age
Non-invasive indexes of fibrosis have been developed and validated mainly in patients between 35 and 65 years of age. In patients over 65 years, a high rate of false positives has been observed for advanced fibrosis, drastically decreasing the NFS and FIB-4 specificity in these patients. McPherson et al. 28 established age-adjusted cut-off values to improve the specificity in these patients. By adopting the revised threshold in patients older than 65 years of age (FIB-4> 2.0 and NFS> 0.12 for the lower cut-offs), the sensitivity and specificity achieved for advanced fibrosis were 77% and 70%; and 80% and 70%, respectively, compared to the previous cut-off values (specificity 20% for NFS> -1.445 and 35% for FIB-4> 1.3). Likewise, non-invasive liver fibrosis scores in adults <35 years and, mainly, in pediatric patients have a low capacity to diagnose significant fibrosis with AUROCs between 0.36-0.6129,30.
Influence of diabetes mellitus
The diagnostic precision of NITs for advanced fibrosis in patients with MALFD and diabetes mellitus is lower than in patients without it. Bertot et al.32 demonstrated a lower AUROC for FIB-4, NFS, and APRI in patients with DM. Also, they observed that up to 21% and 27% of patients with diabetes mellitus, despite showing a low risk according to NITs, suffered ultimately from decompensation and hepatocarcinoma, respectively.
Influence of obesity
Up to 90% of obese patients have MAFLD. Considering this, NITs have not been correctly validated in the obese population despite they have a high risk of fibrosis. In fact, lower diagnostic precision has been observed for FIB-4, APRI, and NFS in obese patients, especially those methods that include BMI in their algorithm33. This parameter does not significantly discriminate the risk of fibrosis and may, therefore, bias the results in obese patients. Joo et al.34 demonstrated significantly higher FIB-4 and NFS AUROCs in non-obese patients compared with the obese subgroup (FIB-4: 0.965 vs. 0.835, and NFS: 0.965 vs. 0.801).
Influence of liver damage
NITs identifying steatosis (FLI, HSI, and SteatoTest®) have been externally validated. However, they are influenced by fibrosis and inflammation. Consequently, they are not able to accurately quantify steatosis in situations of great liver inflammation or advanced fibrosis31.
Undetermined results from NITs
One of the main concerns of NITs is the high percentage of patients located in the grey zone (undetermined results) for advanced fibrosis, representing up to 25-30% of patients for NFS and FIB-435,36. By contrast, HFS, although it maintains a grey zone, is able to reduce significantly the number of patients allocated at risk of indeterminate fibrosis37. In this setting, an alternative for patients with initial results of indeterminate fibrosis is to use more than one NIT to increase the accuracy of the detection of mild or advanced fibrosis27.
Tabla 1
Pros and cons of using NITs to detect advanced fibrosis in MAFLD.
Conclusion
There are a multitude of serum biomarkers and NITs currently accessible for MAFLD. Their usefulness for detecting steatosis and fibrosis is optimal, although the diagnosis of steatohepatitis remains a challenge. The balance between patented and non-patented, as well as the combination of NITs, should represent an interesting approach to overcome the limitations that they show. Therefore, biomarkers and NITs should represent the first step in the diagnosis of MAFLD.

 Descargar número completo
Descargar número completo Download full issue
Download full issue